One Breathe Is All It Takes
Revolutionizing cancer screening through breath Analysis
Pulmostics has created the first of-its-kind non-invasive detection system for Breast Cancer

Breast Cancer
Detecting Breast Cancer can be as simple as exhaling
Breast Cancer has become the #1 most diagnosed cancer worldwide.
70% of all cases are diagnosed at stage 3 or later.
Breast cancer has become the second leading cause of Cancer deaths (behind lung cancer) in women.
Early detection is the key to reducing the cost of treatment, the pain and trauma of treatment and the number of deaths.
Women are not screened due to Cost, Access Discomfort, Social constraints.
Pulmostics Limited
Breath diagnostics for breast cancer can be as simple as 1, 2, 3
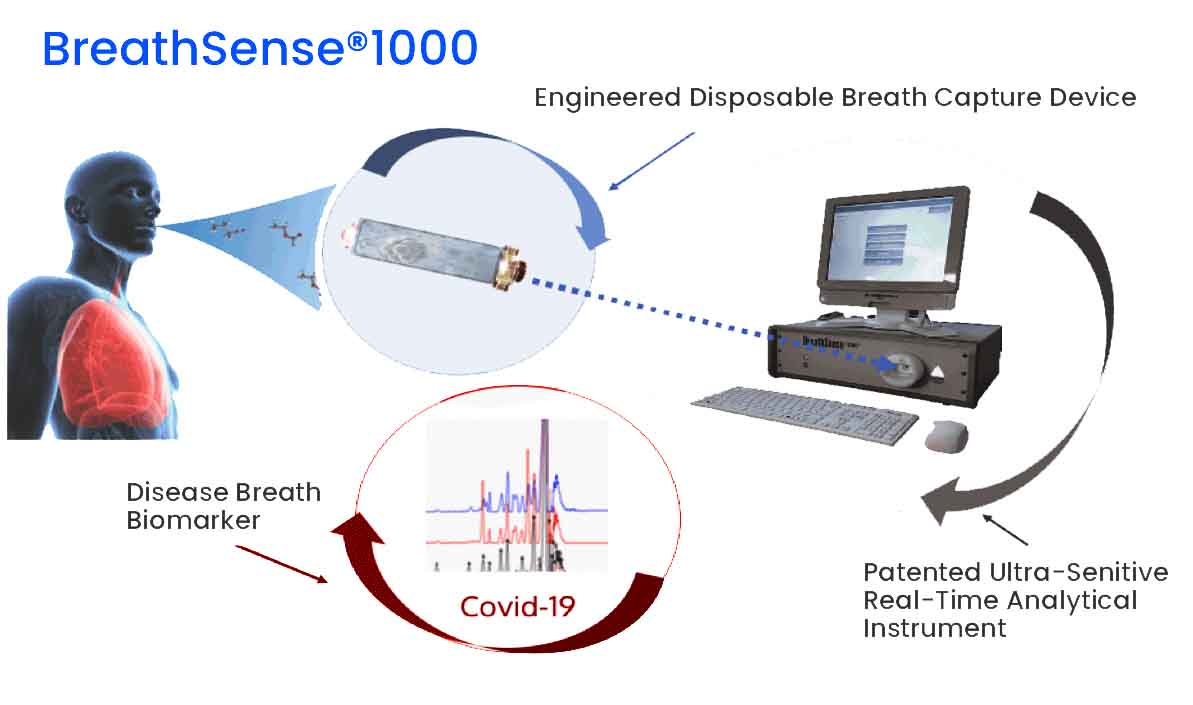
Pulmostics Limited was incorporated in July 2015 for the purpose of developing a diagnostic system that detects early breast cancer based on Breath Analysis. BreathSenseTM 1000, the first of its kind was then born.
Pulmostics has 3 issued and two pending patents. Clinical Trials have already been done with BreathSense technology demonstrating real-time detection of breast cancer with 80% sensitivity and rapid detection of lung cancer with 76% sensitivity. Pulmostics has contracted with Christus Muguerza to conduct a large-scale clinical trial (1350 women) to begin as early as December 2022.
Turnkey Solution
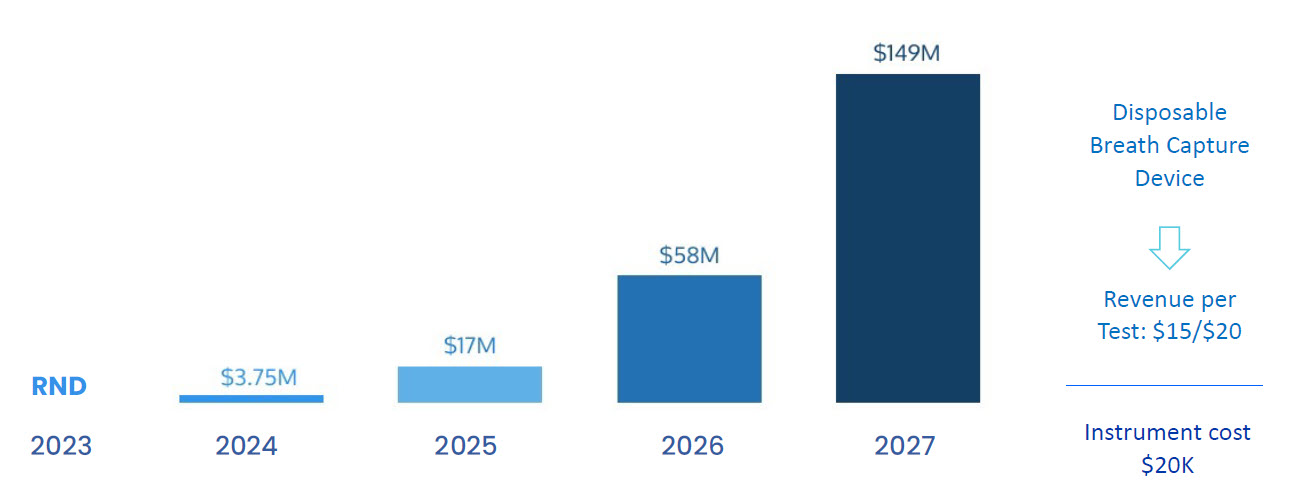
10-20 USD per test
Disposable Breath Capture Device
Desktop Analytical Instrument

Introduction
Breast cancer is the most commonly diagnosed cancer in women, in whom it is second only to lung cancer as a cause of cancer death. The Susan G Korman foundation predicts that more than 287,000 US women will be diagnosed with breast cancer in 2022 and nearly 43,000 will die of the disease.
In order to reduce breast cancer deaths, many countries have established screening mammography programs to detect and treat the disease. However, screening mammography is associated with an increased risk of radiation-induced breast cancer, and with over diagnosis and overtreatment.
These limitations of screening mammography have stimulated the search for new tools to identify early-stage breast cancer without any discomfort or risk.

Annual Global Market
The clinical value of breath testing has been increasingly recognized. The urea breath test is used to detect Helicobacter pylori infection and measurements of nitric oxide concentrations in breath are used to monitor the severity of bronchial asthma.
Pulmostics Breath Sense 1000, a low cost easy non-invasive system to screen for breast cancer through breath analysis.
Annual Global Market
The global breast cancer screening market size was valued at $16.5B in 2022.
The global cancer diagnostics market is estimated to reach $26.6 billion by 2026, growing at a CAGR of 11.5% in that time.
Market Opportunity
Plenty of small organizations are attempting to take advantage of this sector. Breathonix and Owlstone Medical are also private enterprises with similar operations. Breathonix is focused on COVID-19 detection, while Owlstone is concerned with lung cancer.
They currently do not have as wide of a breadth of focus, not to mention the commercial opportunities that Pulmostics have lined up internationally. They are preparing to partner with the largest Catholic healthcare system in Mexico. The Mexican Government employee’s health care provider, and Asia’s largest integrated health care group.
There is an obvious market opportunity with the incidence level of these fatal conditions, the lack of proactive screening options and the absence of dominant competition operating in this space.

Lack Of Competition
Commercial Opportunities

Largest Catholic heath care system in Mexico. Clinical Trial partner 9 hospitals 40 clinics.

Mexican Government employee’s health care provider, 115 hospitals, 6 million covered employees.

Asia’s largest integrated health care group 72 hospitals, 120 primary care clinics and 700 diagnostic centers.
Revenue Projections
Study Collaborative
Christus Muguerza Hospital and Pulmostics Ltd.
Breath and Urine Test as A Screening and Diagnostic Tool for Breast Cancer in Women
1
Stage 1 - Algorithm Development
Patient population:
450 (150 patients per
each of Groups 1 – 3)
Sites: One
Objective: Feasibility test and algorithm development.
Stage 2 - Algorithm Validation
Patient population:
900 (300 patients per
each of Groups 1 – 3)
Sites: One
Objective: Contingent on completion of Stage 1 according to acceptance criteria. Validation of the algorithm developed in stage one in a limited population.
Stage 3 - Efficacy Validation
Patient population:
9,900 (3,300 patients per
each of Groups 1 -3)
Sites: 4-9
Objective: Contingent on successful completion of Stage 2 according to acceptance criteria. Enable assessment of efficacy in represented
population segments with appropriate statistical power.
Background:
The incidence of false positive results associated with x-ray mammography, as confirmed via biopsy, is higher than desired, resulting in higher cost and lower quality of care.
Potential Solution:
Utilization of a breath test as a method for screening for breast cancer in women has demonstrated sensitivity and specificity comparable to mammography in observational studies of small populations as well as lower false positive rate than mammography in women with abnormal mammograms in small studies.
Clinical Operations Plan:
An operating plan will be developed in collaboration with Christus Muguerza in order to capture appropriate procedures and techniques, including Standard Operating Procedures for study site operation.